Certainly, the previous copper wire experiment can be run on a comparative basis in which a first cell would be dosed with a suitable quantity of the Batteryvitamin substance and a second be used as a control.
The appearance of the two copper electrodes at the conclusion of the experiment would reflect the effect of the Batteryvitamin; with the dosed copper having acquired a hazy gray coloration and the control copper having become darker.
However, the experiment takes in the order of 24 hours to complete and the distinction between the two electrodes might not necessarily appear clear cut via the camera. It would therefore be useful to find a way of compressing the experiment's duration to, say, about 4 hours and also to use more convenient materials and simpler test equipment.
In this regard tin appears to be able to provide an ideal substitute for lead for experimental purposes, also being a group IV element, having the same oxidation states of +2 and +4, possessing almost identical electrochemical potentials at Pb + 2e 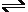 Pb -0.1262 V and Sn + 2e
Pb -0.1262 V and Sn + 2e 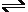 Sn -0.1375 V and having similar ionic radii. Both have high hydrogen overpotentials. Tin is easy to handle and is relatively soluble in sulfuric battery acid and can therefore help to provide an excellent perspective on the actual electroplating process within a comparatively short period of time.
Sn -0.1375 V and having similar ionic radii. Both have high hydrogen overpotentials. Tin is easy to handle and is relatively soluble in sulfuric battery acid and can therefore help to provide an excellent perspective on the actual electroplating process within a comparatively short period of time.
This movie shows a positive electrode consisting of a coil of 97:3 tin-silver solder wire being electroplated onto a negative electrode wire in regular sulfuric battery acid over a period of 8 hours, although 4 hours would have been sufficient. All the metal that can be seen growing on the negative wire originates from the positive. It is much easier to monitor this growth than to measure erosion at the positive electrode.
The extra time provides a magnified representation of the interesting fractal dendritic development typical of plain acid electroplating, closely resembling mossing sometimes found in lead-acid battery cells, for example, after a period of 5 years. (In commercial electroplating a smooth surface can only be obtained by including metal salts and grain refiners in the electrolyte.)
TEST EQUIPMENT
Regular 12V automotive battery, for use as power supply;
150 ohm, 2 watt resistor, as current regulator.
